O-Level Chemistry: Types of Chemical Reactions
1. Combination Reaction
Definition: Two or more reactants combine to form a single product.
General Formula: A + B → AB
- 2H₂ + O₂ → 2H₂O
- N₂ + 3H₂ → 2NH₃
2. Decomposition Reaction
Definition: A single compound breaks down into two or more simpler substances.
General Formula: AB → A + B
- 2H₂O → 2H₂ + O₂
- CaCO₃ → CaO + CO₂
3. Single Displacement (Substitution) Reaction
Definition: An element reacts with a compound, displacing another element.
Formula: A + BC → AC + B
- Zn + 2HCl → ZnCl₂ + H₂
- Cl₂ + 2NaBr → 2NaCl + Br₂
4. Double Displacement (Metathesis) Reaction
Definition: Exchange of ions between two compounds to form two new compounds.
Formula: AB + CD → AD + CB
- Na₂SO₄ + BaCl₂ → BaSO₄↓ + 2NaCl
- AgNO₃ + NaCl → AgCl↓ + NaNO₃
5. Combustion Reaction
Definition: A substance reacts with oxygen, releasing heat and light.
Formula: Fuel + O₂ → CO₂ + H₂O (for hydrocarbons)
- CH₄ + 2O₂ → CO₂ + 2H₂O
- C + O₂ → CO₂
6. Redox Reaction
Definition: Reactions in which oxidation and reduction occur simultaneously.
- Zn + CuSO₄ → ZnSO₄ + Cu
- 2Na + Cl₂ → 2NaCl
7. Neutralization Reaction
Definition: Acid reacts with a base to form salt and water.
Formula: Acid + Base → Salt + Water
- HCl + NaOH → NaCl + H₂O
- H₂SO₄ + 2KOH → K₂SO₄ + 2H₂O
8. Precipitation Reaction
Definition: Two aqueous solutions react to form an insoluble solid called a precipitate.
- BaCl₂(aq) + Na₂SO₄(aq) → BaSO₄↓ + 2NaCl
9. Displacement Reaction
Definition: A single element is simultaneously oxidized and reduced.
- 2H₂O₂ → 2H₂O + O₂
10. Combustion of Metals
Definition: Some metals react with oxygen to form metal oxides.
- 4Al + 3O₂ → 2Al₂O₃
- 2Mg + O₂ → 2MgO
11. Other Important Notes
- Reaction conditions like heat, light, and catalysts often influence reaction types.
- Most reactions can be classified broadly into redox, acid-base, combination, decomposition, or displacement.
- Some reactions fit into multiple categories (e.g., combustion is also a redox reaction).
- Always balance chemical equations before studying reaction types.
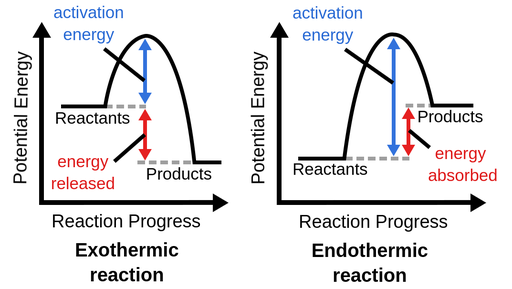
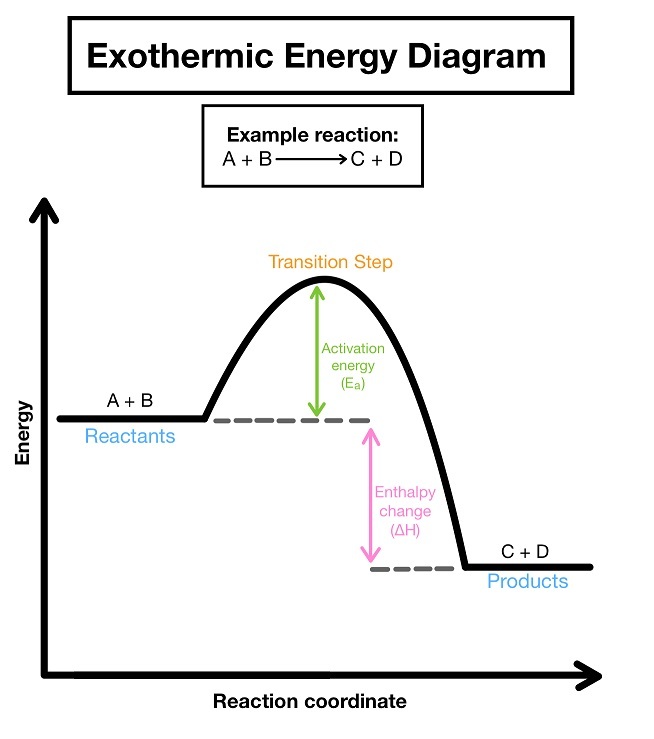
12. Exothermic Reaction
An exothermic reaction releases energy to the surroundings. Energy of reactants > products, heat change negative (-).
Ca + H₂O → Ca(OH)₂
CO₂ + H₂O → H₂CO₃
13. Endothermic Reaction
An endothermic reaction absorbs energy from the surroundings. Energy of products > reactants, heat change positive (+).
CaCO₃(s) → CaO(s) + CO₂(g)